Determination of the acid neutralization capacity (ANC)
Jutta Laine-Ylijoki, Markku Juvankoski, Tommi Kaartinen, Elina Merta, Ulla-Maija Mroueh, Jarno Mäkinen, Emma Niemeläinen, Henna Punkkinen & Margareta Wahlström, VTT Technical Research Centre of Finland Ltd, P.O. Box 1000, FI-02044 VTT, Finland.
Introduction
Acid neutralization capacity (ANC) -property gives information on the behaviour of the waste material itself, but it is also important for understanding the influence on the surroundings at a site (e.g. due to interaction with other materials or leachates).
The overall waste body at a site needs to become and remain roughly neutral (pH 7-8) in order to avoid any long-term adverse effects in the waste body or in the environment of a site. Acidic or alkaline leachate occurs when the neutralization effects (e.g. minerals) are not able to neutralize acid or base generated by reactions in waste (e.g. sulphide oxidation, anaerobic or aerobic digestion of organics) or by external effects (e.g. acid rain, carbonation due to reaction with atmospheric carbon dioxide). Adverse effects (e.g. increase in metals and organics leachability, negative impact on landfill barriers) may be caused both by low or high pH values in waste leachate.
The buffer capacity of a waste can be measured and expressed in terms of the so-called acid neutralization capacity (ANC). It is usually not possible to assess the buffering capacity of an entire site and therefore the pH stability of a site is evaluated through the testing of single waste materials and taking into account the actual site conditions in the test interpretation (e.g. waste streams, site construction, management etc). Therefore, the ANC property cannot be evaluated alone. For evaluation of risk related to specific ANC data, information is needed on the waste properties that are critical to pH behaviour. (Wahlström et al. 2009)
ANC as a parameter
The acid neutralization capacity (ANC) of waste is a measure for the overall buffering capacity against the change in pH which might stem from reactions in the waste materials themselves or external conditions. In both cases, this leads to a change of pH in site leachate (e.g. through formation of acid). ANC measurements cover both the acid neutralization capacity of the waste solution and the solids present.
The term “alkalinity” is often incorrectly used for ANC. Alkalinity means the ability of a solution to neutralize acids to the equivalence points of carbonate or bicarbonate. According to a more general definition given in the IPCC report (2001), alkalinity is a measure of the capacity of water to neutralize acids. The data can also be used to assess the effect of external influences, such as carbonation and oxidation (acid production resulting from sulphide oxidation in sulphide-containing waste, e.g. mining waste or acidic sulphate soils) on the pH of a waste. The external influences need to be calculated in the same units to allow comparison, and therefore carbonation would have to be assessed through the volume of gas, concentration of CO2, and conversion to corresponding moles of H+ neutralizing capacity. The terms often used in connection with acid neutralization capacity are collated in Table 1. (Wahlström et al. 2009)
Table 1. The terms often used in connection with acid neutralization capacity (Wahlström et al. 2009).
| Matrix | Term | Reference | Definition |
| Waste | Acid neutralization capacity (ANC) | CEN/TS 15364: Characterization of waste – Leaching behaviour tests – Acid and base neutralization capacity test | The acid and/or base neutralization capacity of a material |
| Waste from extractive waste | Acid potential (AP) | EN 15875: Characterization of waste – Static test for determination of acid potential of sulphidic waste | The maximum potential acid generation from a sample assuming that all sulphur occurs as pyrite and that acidity will result from its complete oxidation |
| Neutralization potential (NP) | The capacity of a sample to neutralize the generated acidity | ||
| Net neutralization potential (NNP) | Difference of acid potential and neutralization potential NNP = NP – AP | ||
| Neutralization potential ratio (NPR) | Ratio of neutralization potential and acid potential NPR = NP / AP | ||
| Soil – liming | Liming effects | ISO 10693: Soil quality – Determination of carbonate content – Volumetric method | |
| Cement-stabilized material | Acid neutralization capacity (ANC) | Stegemann (2002) | The ability of a solidified product to neutralize acid |
Description of the test methods
The acid neutralizing capacity is often defined as a measure of the amount of base present that can accept hydrogen ions from a strong acid. Since the extent of these acid base reactions is dependent on pH, the ANC is a function of pH. However, a single value of ANC is often reported by choosing a specific pH for determination. Very often a curve of the ANC characteristics for a specific waste is presented.
There are different waste test methods for determination of the acid neutralization capacity of a waste material. The ANC is generally determined through titration to a selected pH value. The property is expressed in mol/kg. In waste characterization, ANC testing not only covers acid neutralization capacity but also base neutralization capacity (BNC). Such ANC/BNC characterization test data can be used to assess the amount of acid (as H+) or base (OH–) needed to reach a given final pH in a waste.
In the laboratory, ANC is usually determined by digesting a sample in a solution containing acid. Acid consumed in fixed testing conditions represents the ANC of a sample. Tables 2 and 3 show the basic principles of tests developed to measure the ANC of waste materials.
Table 2. European test methods developed for determination of the neutralization potential of waste (Wahlström et al. 2009).
| Test | pH-dependence test CEN/TS 14997 | pH-dependence test CEN/TS 14429 | Static test EN 15875 |
|---|---|---|---|
| Test level | Basic characterization | Basic characterization | Screening / compliance testing |
| Scope | Primarily influence of pH on the leachability of inorganic constituents from waste materials | The potential of sulphide-bearing materials to form acidic drainage | |
| Developed for (materials) | All waste materials | All waste materials | Sulphidic extractive waste |
| Principle | Sample is mixed with distilled water and pH level of the solution is kept at a predetermined pH value by using an automated pH titrator | Sample is mixed with leachant containing pre-selected amounts of acid or base in order to reach stationary pH values at the end of the extraction period | Digestion of sample with acid. Acid is back-titrated to 8.3 |
| Sample amount | 15, 30 or 60 g | 15, 30 or 60 g | 2 g |
| Sample grain size | < 1 mm | < 1mm | < 0.125 mm |
| Target pH | Each pH value tested separately. Typically pH range 3-12 covered (to be selected based on material and disposal conditions) | Each pH value tested separately. Typically pH range 3-12 covered (to be selected based on material and disposal conditions) | Digestion around pH 2, back titration of excess acid to pH 8.3 (digestion pH only roughly controlled) |
| L/S | Target 10 | Target 10 | 45 plus acid added |
| Test duration | 48 h | 48 h | 24 h |
| Requirement on achievement of equilibrium condition in test | yes | yes | no |
| Test limitations | Time-dependent leaching of substances (slowly leachable) not addressed; matrix interference | Time dependent leaching of substances (slowly leachable) not addressed; matrix interference | Only fast reactive compounds included; relevance of grain size requirement for waste rock |
| Expression of results | Acid/base added for each target pH; concentrations of analyzed substance in test eluates for each target pH | Acid/base added for each target pH; concentrations of analyzed substance in test eluates for each target pH | Calculation of NPR ratio (the ratio between NP and AP) |
| Criteria for test interpretation | No | No | EU criteria for inert mine waste classification (Commission Decision 2009); indicative NPR values for identification of risk of formation of acid drainage |
Table 3. Description of European test methods developed for determination of the neutralization potential of waste (Technical Committee CEN/TC 292 2012).
| CEN/TS 14997 with continuous pH control | In CEN/TS 14997 test sample (15, 30 or 60 g) is mixed with distilled water for 48 hours at an L/S ratio of about 10. The pH level of the mixture is kept at the predetermined pH value by using an automated pH titrator. An equilibrium is established as a result of continuous adjustment of pH. Size reduction (<1 mm) of the sample is performed to accelerate reaching of equilibrium. The acid or base consumption recorded by a titrator shows the ANC or BNC at a given pH value. |
| CEN/TS 14429 with initial acid/base addition | Separate test portions are leached at a fixed L/S ratio with leachants containing pre-selected amounts of acid or base in order to reach stationary pH values at the end of the extraction period. Each leachant is added in three steps in the beginning of the test. At least 8 final pH values are required, covering at the minimum the range of pH 4 to pH 12 (both included, i.e. the lowest value 4 and the highest value 12). The amounts of acid or base needed to cover the pH range can be derived from the results of a preliminary titration, from available experimental data on the material to be tested or from an arbitrary division of the predetermined maximum consumption of acid and base. The tests are carried out at a fixed contact time at the end of which equilibrium condition can be assumed to be reached for most constituents in most waste materials to be characterized. |
| Static test EN 15875 | In the static test EN 15875 2 grams of finely ground sample is digested for 24 hours in 90 ml of HCl solution. The test aims at final digestion pH of 2.0-2.5 and the amount of 1 M HCl added to the solution in the early stages of the test is determined on the basis of the carbonate content. After digestion the solution is back titrated to pH 8.3 to measure the amount of acid left in the solution. The acid consumption in the test is converted to the neutralization potential (ANC) of a sample. |
Comparison of European methods for ANC testing
A comparison of ANC results for a mining waste (tailings) gained with different test methods is shown in the Figure 1. The results from CEN/TS 14429 and CEN/TS 14997 are almost similar. The static test seems to give lower values for ANC. This can probably be explained by the nature of the tests. In the static test prEN 15875 only three time-points to add acid to the mixture exist. Between the acid additions the pH of the mixture climbs thus preventing the buffering of some minerals that are only active at lower pH levels.
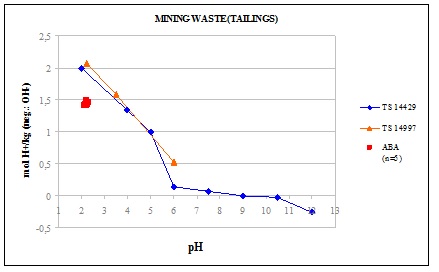
Figure 1. Example of ANC for mining waste with different tests. ABA test corresponds to the static test. (Wahlström et al 2009)
Other supplementary approaches
The release of contaminants from a solid material is often controlled by (geo)chemical mechanisms, such as solubility of minerals/salts, sorption to reactive mineral and organic surfaces and complexation to (in)organic ligands. These fundamental processes dictating the leaching can be modelled using appropriate leaching data and speciation models. For the production of data to be used in hydrogeological modelling, a widening of the pH range will be useful (e.g. pH = 1 to 13).
The ANC results from pH-dependence testing (and also static test) often correlate with the dissolution of calcium-containing minerals or materials. Secondary minerals, which may precipitate and later re-dissolve, are important to the neutralizing processes (Yan et al. 1998). Ashes and slag also consist of a considerable amount of amorphous phases that also contribute to ANC, which are difficult to evaluate through short-time batch testing. The understanding of secondary minerals and amorphous phases can be evaluated through geochemical modelling.
The effective NP of mine waste can also be calculated based on the mineralogical composition of a sample and relative mineral reactivity of minerals. Different minerals can neutralize acid drainage at different rates and in different pH ranges. The test conditions of widely used laboratory procedures to determine NP do not distinguish between such differences and overestimation of NP can often result. The calculation method is based on the determination of the major rock-forming elements of a sample, usually referred to as whole rock analysis, in combination with the analysis of inorganic carbon (carbonate), as inputs to the calculations. A major advantage of this approach is that whole rock analyses are often already available, having been determined as part of the exploration and ore-mapping process. The procedure is considered to be a cost-effective means of providing confident routine ARD prediction when used in combination with other tests and analyses. (Lawrence & Scheske 1997, Packtunc 1999).
Evaluation of ANC results
In the ANC testing of waste, the ability of a material to neutralize acid or base is evaluated at a selected pH value. Both the neutralization potential and acid potential are of importance in the evaluation of ANC properties. Generally, in ANC testing more focus is given to the neutralization potential than to the acid potential of the waste. A special case is always waste subjected to carbonation (here pH change to pH around 8 will occur independent of the neutralization potential values).
The real effects on-site including the external effects need to be assessed by testing weathered samples from the field or by monitoring of the actual pH in the landfill leachate. The evaluation of a given neutralization potential depends on the longer term conditions under which the waste will be placed and on whether the potential acidification is inherent in the waste (i.e. whether it has an acid-producing capacity) or whether the pH change will occur only through external influences.
The critical value for the neutralization potential depends on the case in question. No clear indications of critical values for the neutralization potential are presented in the literature. Moreover, the neutralization potential values are also pH-dependent. Calcite (CaCO3), having a neutralization potential of 20 mol H+/kg, is a fast reactive mineral and usually indicates an upper limit for the neutralization potential. The values for the slow weathering minerals (with low practical meaning in neutralization) lie around 0.2 mol H+/kg at pH 4-5. (Jambor 2005, INAP 2009, Wahlström et al. 2009)
It is also shown that the sulphide content of 0.1 weight % (which is also the limit for inert waste requiring no further ARD testing) creates an acid potential (and thus also a demand for the neutralization potential) of 0.6 mol H+/kg. Based on the observations above, an indication of a low and critical NP value is here chosen as 0.2 mol H+/kg. Actually, also a value of 0.6 mol H+/kg could be argued as low based on acid potential created from a sulphide content of 0.1 %. From a practical point of view in testing, a value of 3-4 mol/kg at pH 4-5 can be seen as an example of high neutralization capacity. (Wahlström et al. 2009)
References
INAP 2009. The GARD Guide. The Global Acid Rock Drainage Guide. The International Network for Acid Prevention (INAP). Available at: http://www.gardguide.com
Jambor, J.L., Dutrizac, J.E. & Raudsepp, M. 2005. Neutralization potentials of some common and uncommon rocks, and some pitfalls in NP measurements. 12th Annual British Columbia MEND ML/ARD Workshop, 30.11.–1.12.2005.
Lawrence, R.W. & Scheske, M. 1997. A method to calculate the neutralization potential of mining wastes. Environmental Geology 32(2), September 1997.
Paktunc, A.D. 1999. Discussion of “A method to calculate the neutralization potential of mining wastes” by Lawrence and Scheske. Environmental Geology 38 (1) June 1999.
Stegemann, J. & Buenfeld N.R. 2002. Prediction of leachate pH for cement paste containing pure metal compounds. Journal of Hazardous Materials B90 (2002) 169–188Boels, D. Technical Committee CEN/TC 292 2012. Characterization of waste – Overall guidance document for characterization of wastes from extractive industries. CEN/TR 16376:2012.
Wahlström, M., Laine-Ylijoki, J. Kaartinen, T., Hjelmar, O. & Bendz, D. 2009. Acid neutralization capacity of waste – specification of requirement stated in landfill regulations, TemaNord 2009:580.
Yan, J., C. Baverman, L. Moreno & I. Neretnieks, 1998. Evaluation of the time-dependent neutralising behaviours of MSWI bottom ash and steel slag. Science of the Total Environment, 1998. 216(1-2): p. 41-54.

Leave A Comment
You must be logged in to post a comment.